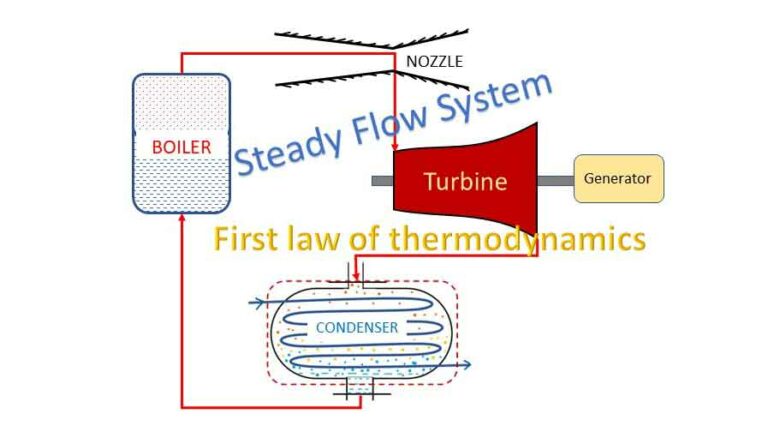
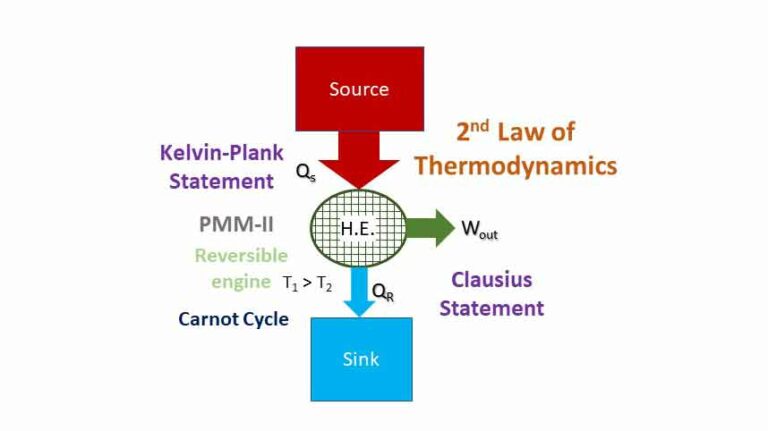
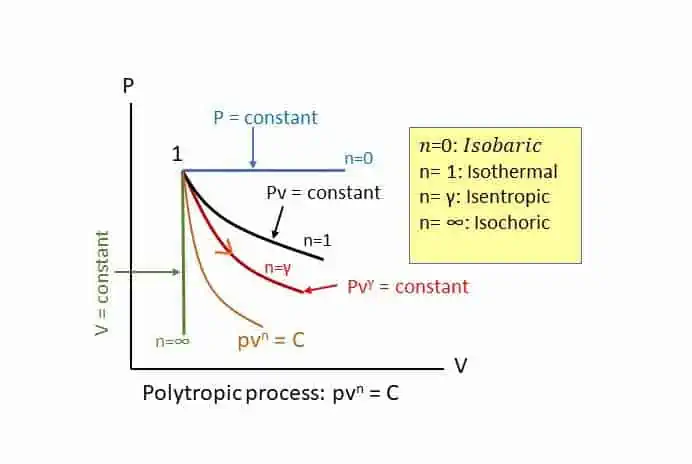
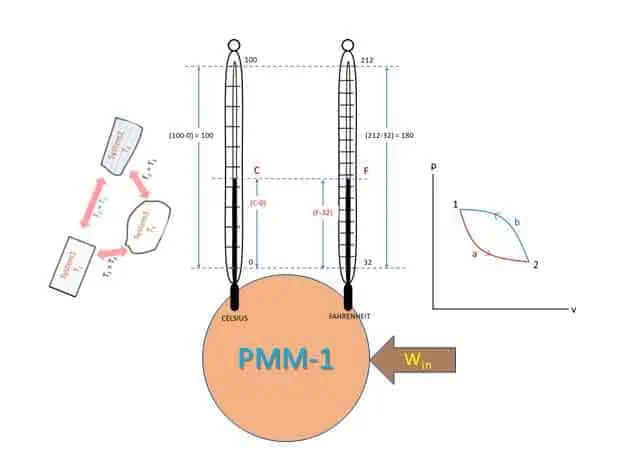
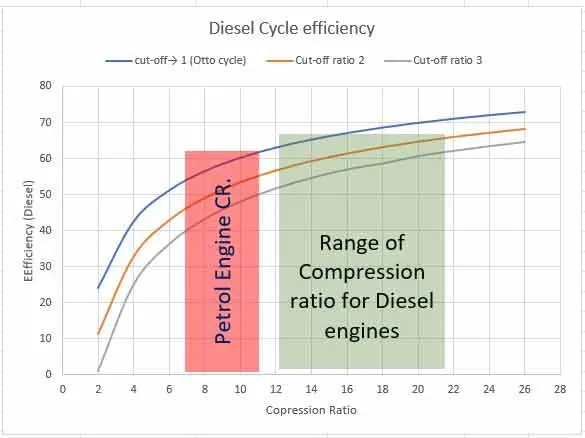
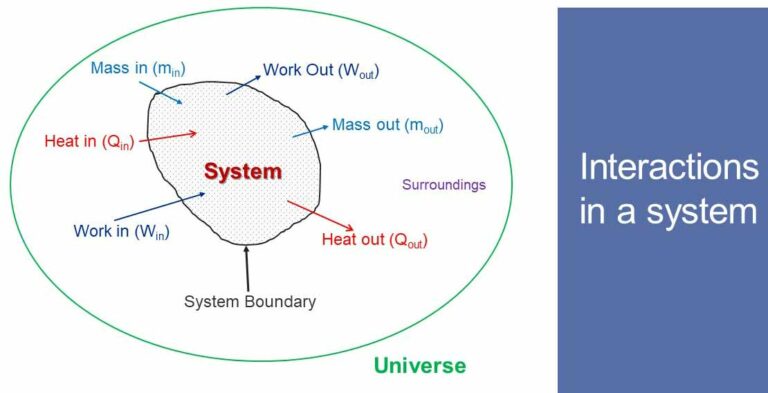
Steady flow system and first law of thermodynamics
In a steady flow process a fluid flow steadily through a control volume. At any point, fluid properties remain constant during the process. There are many engineering equipments which function as steady flow system.