Thermodynamic system
Thermodynamics is a branch of physical science. It deals with various forms of energies including Heat and Work. Energy and Thermodynamic system are particularly important concepts in this branch of science.
According to Donald Haynie, an American biophysicist the term thermodynamics was coined in 1840 from the Greek words therme (heat) dynamis (power). According to Pierre Perrot, the term thermodynamics was used by James Joule in 1858 to explain the relationship between heat and power. Thermodynamics developed in 19th century while scientists were building and operating steam engines. Thermodynamics deals with work and energy of a system and their interactions.
Thermodynamic System
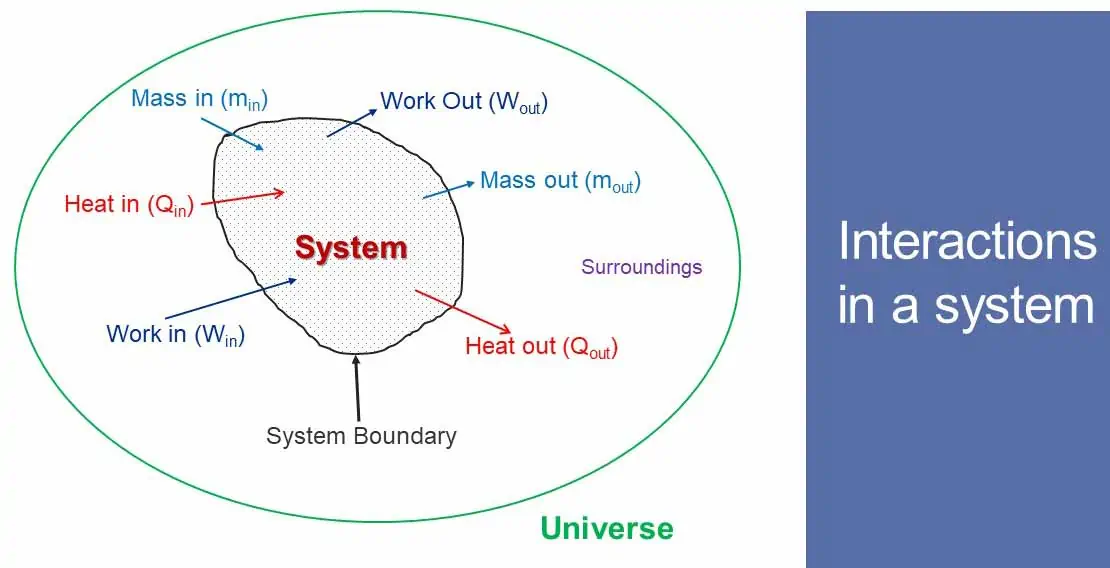
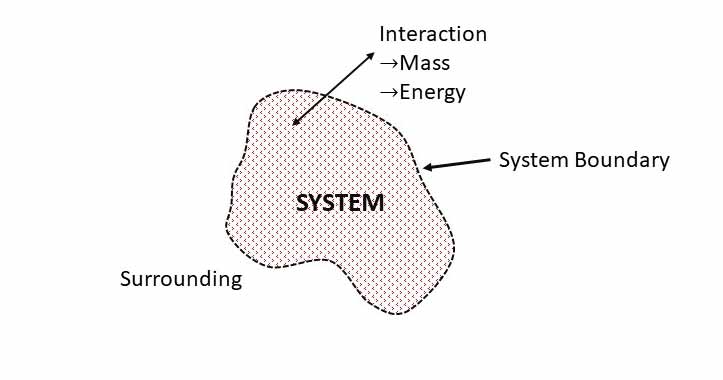
A Thermodynamic system is a quantity of matter, or a region selected for study and separated from everything by a boundary. So, a system is always associated with a system boundary. A boundary separates the system from its surroundings. Any mass or region outside the system is called surrounding.
System Boundary
A system boundary may not be always physical boundary. System boundary has no mass and thickness. It occupies no space. System boundary may be fixed or moving, rigid or flexible and real or imaginary. Mass or energy may or may not move through it.
System Surrounding
Everything outside the boundary that influence the system is called Surrounding. Surrounding can affects the system. System interacts with the surrounding. Movement of mass, energy or both is called interaction of system with the surrounding. So, interaction takes place in the form of Mass transfer, Energy transfer or both.
Types of Thermodynamic system
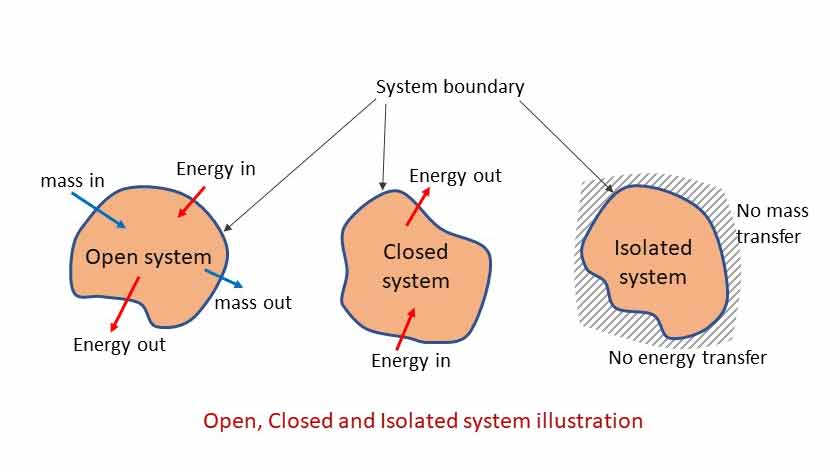
System may be of different types. Depending on the interaction with the surrounding, there are three types of systems. System types depends on the type of the System boundary – whether the boundary allow the mass and energy to move through it or not.
Open System (Control Volume)
Open system allows both mass and energy to interact with the surrounding. Mass as well as energy can flow in and out of the system. Heat is a transient form of energy which crosses the boundary by virtue of temperature difference. Work is another transient form of energy which crosses the boundary by virtue of force.
Closed System (Control Mass)
Closed system does not exchange mass but energy with its surrounding. Only energy can cross the system boundary (inflow or out flow).
Isolated System
The system is isolated from or uninfluenced by the surrounding. Isolated system does not interact with other system or surroundings. Both mass and energy remain constant. Neither mass nor energy can cross the system boundary. Isolated system remains uninfluenced by the surrounding. In practice there is no such system that is not affected by its surrounding. Universe may be an example of isolated system if there is no other universe as its surrounding.
You can watch the following video also
Practical Examples of different Thermodynamic systems:
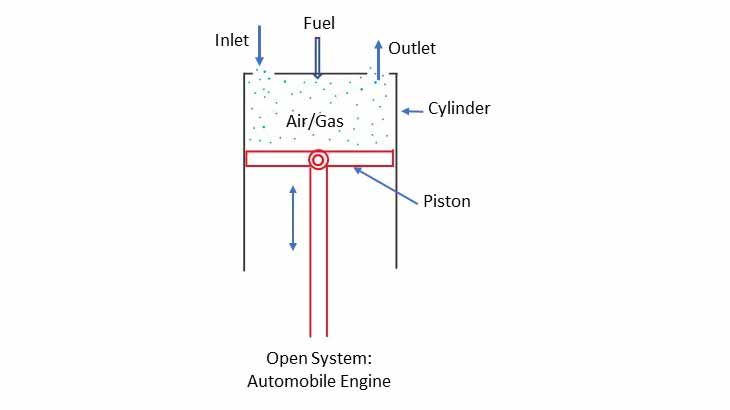
Open System
A piston-cylinder arrangement with inlet and outlet for air/gas is an example of open system. The piston and cylinder walls are the system boundaries. Mass of air and fuel enter the system (mass inflow) and heat is generated due to burning of fuel inside the cylinder (considered as Heat transfer). The High pressure inside the cylinder pushes the piston downwards (Work transfer). The product of combustion goes out to the atmosphere (surrounding) through the outlet valve (mass and heat out flow).
Similarly, in a turbine kinetic/pressure/internal energy of an incoming fluid is converted to mechanical work. In a compressor/pump the kinetic/pressure energy of a fluid is increased by work input to the system. In a boiler (steam generator) heated is added to the system to receive high pressure high temperature steam at the outlet. Nozzle is an example of flow process or open system in which pressure energy of the fluid is converted to kinetic energy.
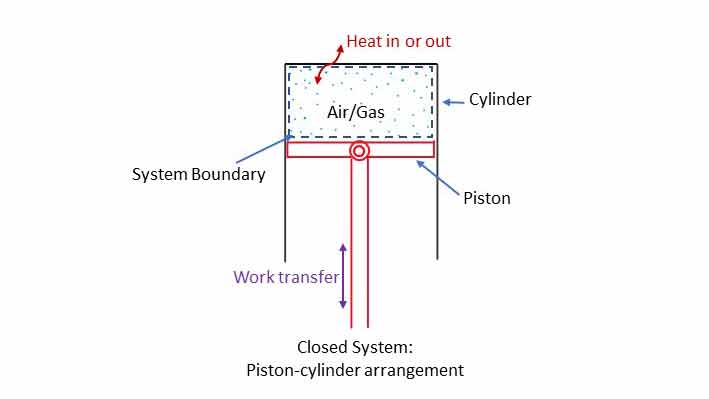
Closed system
A piston-cylinder arrangement with no inlet and outlet for air/gas as shown in the diagram is an example of closed system (no mass flow of fixed mass). If heat is added to the system (energy inflow), air inside will expand in piston will move down (work transfer/energy outflow). If the piston is pushed upward by application of force (work in) the air will be compressed to increase the temperature and the heat may flow out if the cylinder wall is good conductor of heat.
Similarly, a car battery is an example of a closed system with no mass flow.
Isolated system
A heat insulated container like Themo flask in which no heat/ work and mass transfer is possible is an example of an isolated system (fixed mass and energy system). You can wach this short video for some practical examples of thermodynamic system.
Property of a Thermodynamic System
Any object can be described by attributes like weight, mass, volume, colour, shape, size, etc. How can we describe a thermodynamic system? Some parameters or attributes are used to describe a system. These parameters can be measured by instruments, or their values can be calculated. These parameters that describe the characteristics of a system are called properties of a system. Any characteristic or quality of a system is called property.
Intensive property
Properties that are independent of mass of the system are called INTENSIVE PROPERTIES. Intensive property does not change with the mass or size of the system contents. Examples: temperature (T), pressure (P), density etc.
Extensive Property
Extensive property is dependent on mass of the system. The value of the property is sum of the value of the parts of the system. Extensive property is additive by nature. Example volume(V), mass(m), weight(W), Internal Energy(U), Enthalpy(H), Heat Capacity(C), Entropy(S), etc.
Specific Property
It is a special case of intensive property. Expensive property per unit mass is called specific property. For example, volume is dependent on the mass or size, so it is an extensive property of the system. But specific volume (volume per unit mass, v = V/m) is independent of mass or size, so specific volume is a special case of intensive property. Similarly, Specific weight (w = W/m), specific integral energy (u = U/m), specific Enthalpy (h = H/m), Specific Entropy (s = S/m) etc.
State of a Thermodynamic System
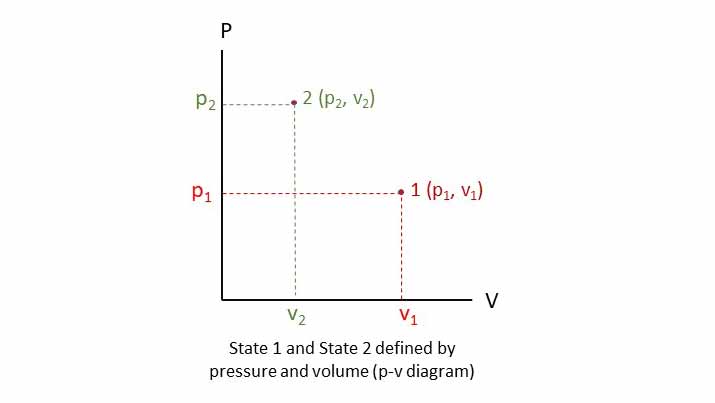
State is the condition of a system at an instant described by its properties. The condition of a system can be defined or described by a set of properties. The properties of a system keep on changing with time and other conditions. So, state of a system (set of properties) changes with time. Change of state means change in properties also.
A state can be defined by a set of properties of the system only. And this set has a unique value for that state only. State and property cannot be defined without each other.
Thermodynamic Equilibrium
Thermodynamic equilibrium is a state of balanced condition. In a state of equilibrium, there is no driving force within the system. So, there would not be any change of state on its own. If there is any change of state, there must be some external force from surrounding or other system acting on the system. A system will be in a state of thermodynamic equilibrium with the surrounding if the following three equilibrium conditions are fulfilled:
To summarise, in thermodynamic equilibrium, pressure, temperature, chemical potential, electrical potential, etc. will be same throughout the system, but shall have the same value as that of its surrounding.
Thermodynamic Process and path
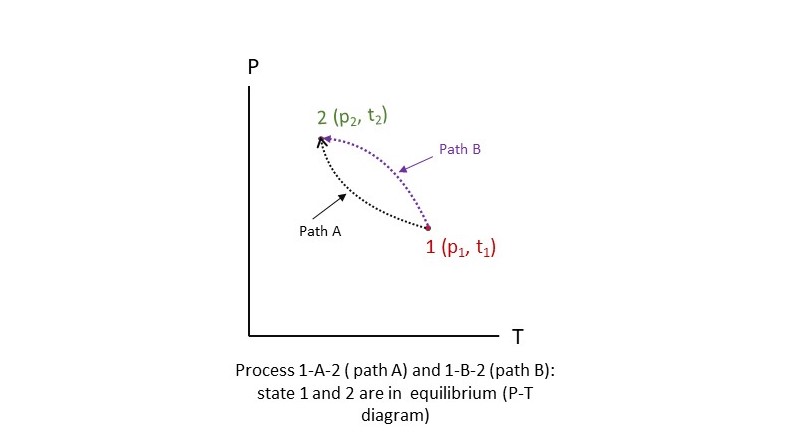
If the state of a system changes from one equilibrium state to another equilibrium state, it is called a process. There are many states within the two equilibrium states of a process. The series of states that a system goes during the process is called path of the process. Figure shows that process 1-2 can be achived by two different paths: path 1-A-2 and path 1-B-2. In both the cases initial and final states are in equilibrium.
Basically, there are two types of processes.
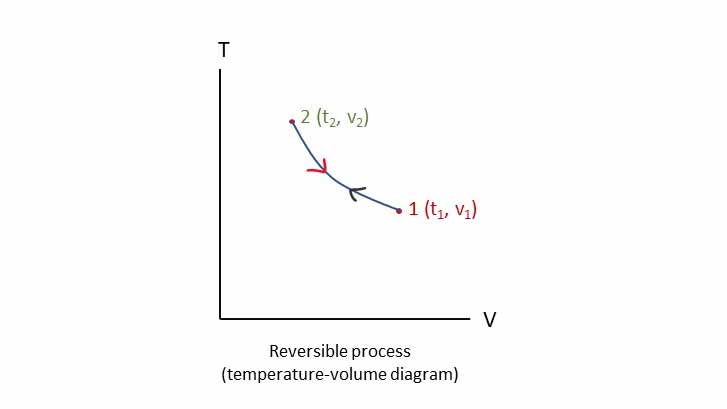
Reversible process:
A reversible process is process which can be reversed or the system and surrounding can be returned to the original state from the final state. Reversible process is an ideal process which cannot occur in nature. Figure shows how the process 1-2 retraces the same reverse path 2-1.
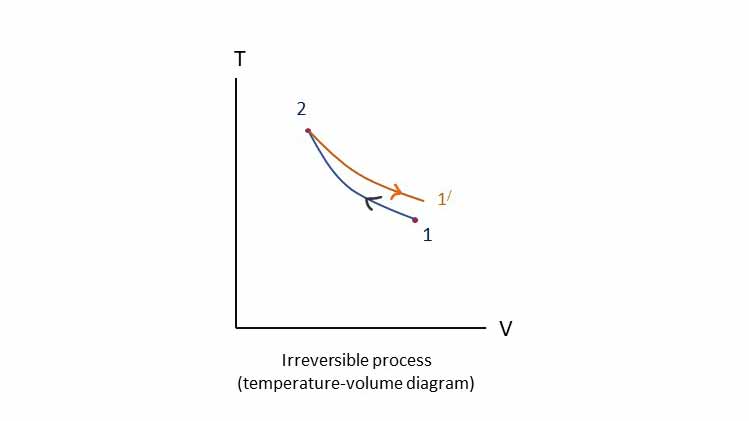
Irreversible process:
If an irreversible process is reversed, the path is not retraced to restore the original state. As shown in the figure process 1-2 when reversed, it traces the the path 2-1/ instead of 2-1.
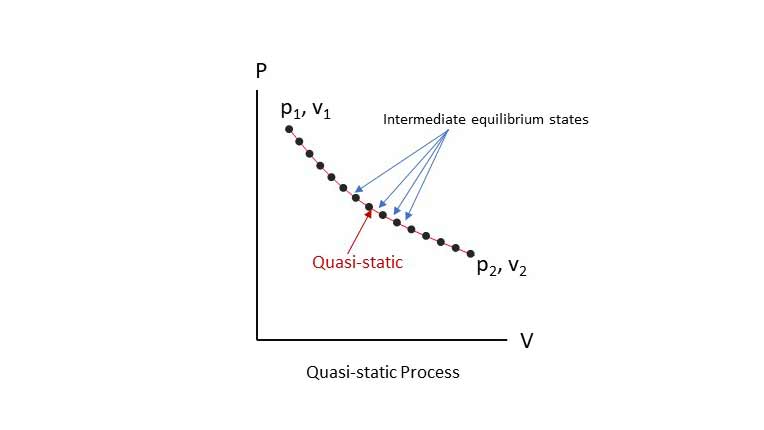
Quasi-equilibrium Process or Quasi-static Process:
When a process undergoes change of state in such a way that at every intermediate state it deviates from the equilibrium infinitesimally (infinitely close to equilibrium) throughout the process, it is called Quasi-static or Quasi-equilibrium process. A Quasi-static process takes place very slowly for the system to remain in internal physical thermodynamic equilibrium. I recomend this video for this sub-topic.
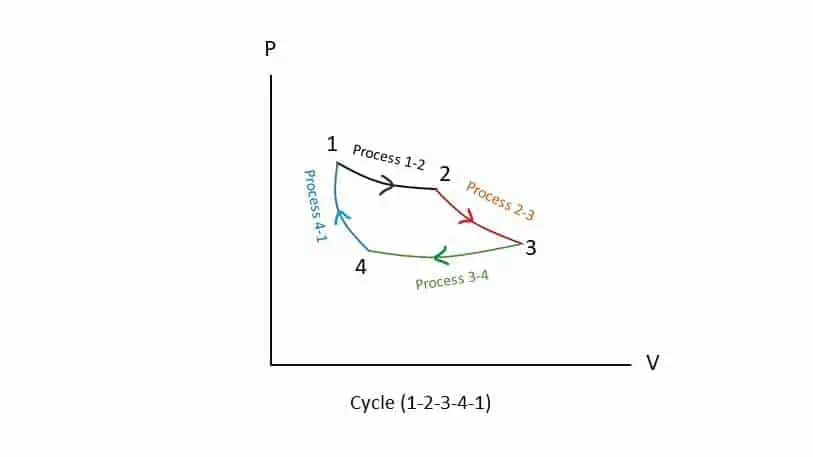
Thermodynamic Cycle
Cycle or Cyclic process is a process or combination of processes such that the initial state is same as that of the final state. In the diagram, process 1-2, process 2-3, process 3-4 and process 4-1 constitute a cycle.
Important Questions and Answers
1 Define a Thermodynamic system.
A thermodynamic system may be defined as a selected area or mass within a boundary separated from and interacting with the surrounding.
2 What are the characteristics of system boundary?
3 What is Control Volume and Control mass?
- If analysis of a system is done over a region of the space, the system is called control volume and popularly known as open system. Mass as well as energy can cross the boundary (mass and energy transfer possible)
- In Control mass, the thermodynamic analysis is carried out on a fixed amount of mass or matter. Mass transfer is not allowed but energy transfer in the form of Heat and/or Work takes place. Such system is popularly known as Closed system.
4 What are the differences between extensive and intensive properties?
| Extensive Properties | Intensive properties |
| Dependent on the mass of a system | Independent of the mass of a system |
| Additive | Not additive |
| Its value for an overall system is the sum of its values for the parts into which the system is divided | Its value remains the same for whole system or only a part of it. |
| Ex: Mass(m), volume(V), Energy(E), Enthalpy(H) etc. | Example: pressure(p), temperature(T) etc. |
5 Compare Process and path of a system.
| Path | Process |
| The succession of states passed through during a change of state forms the path of the change of state. | When a path is completely specified then the change of state is called a process. |
6 Reversible processes are Quasi-static or Quasi-equilibrium: Elaborate the statement.
- After completion of the process, both the system and surrounding may be restored to their initial state without producing any changes in rest of the Universe.
- Carried out infinitely slowly with an infinitesimal gradient (Quasi-static).
- The process can be executed in either of the direction.
- No loss such as friction etc are present.
- Heat and work interactions in the reverse process are equal and opposite in direction to that of the forward process.